|
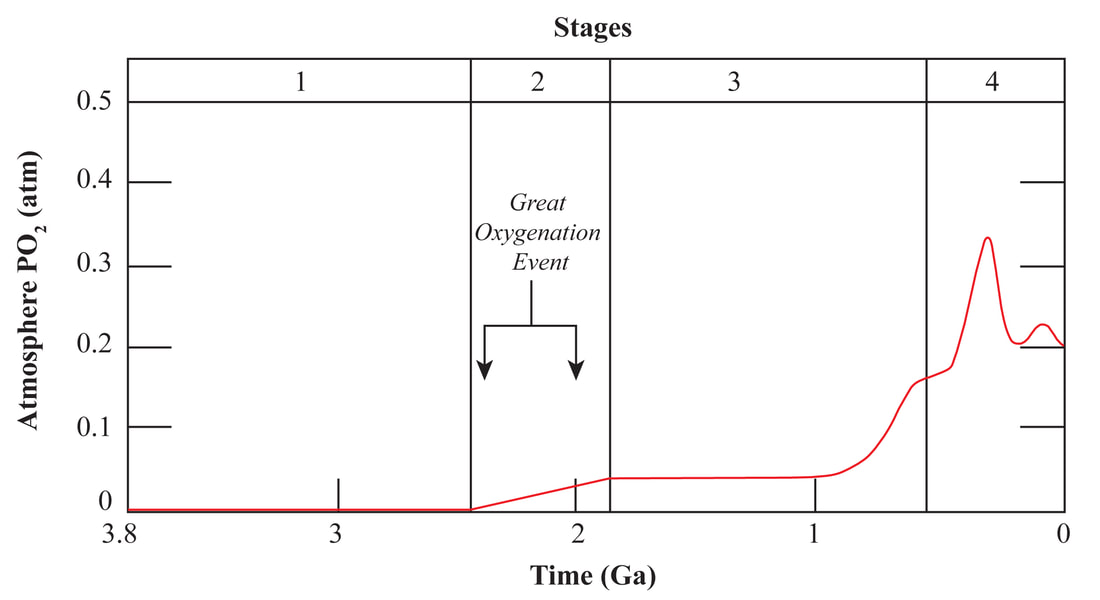
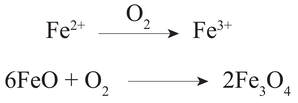
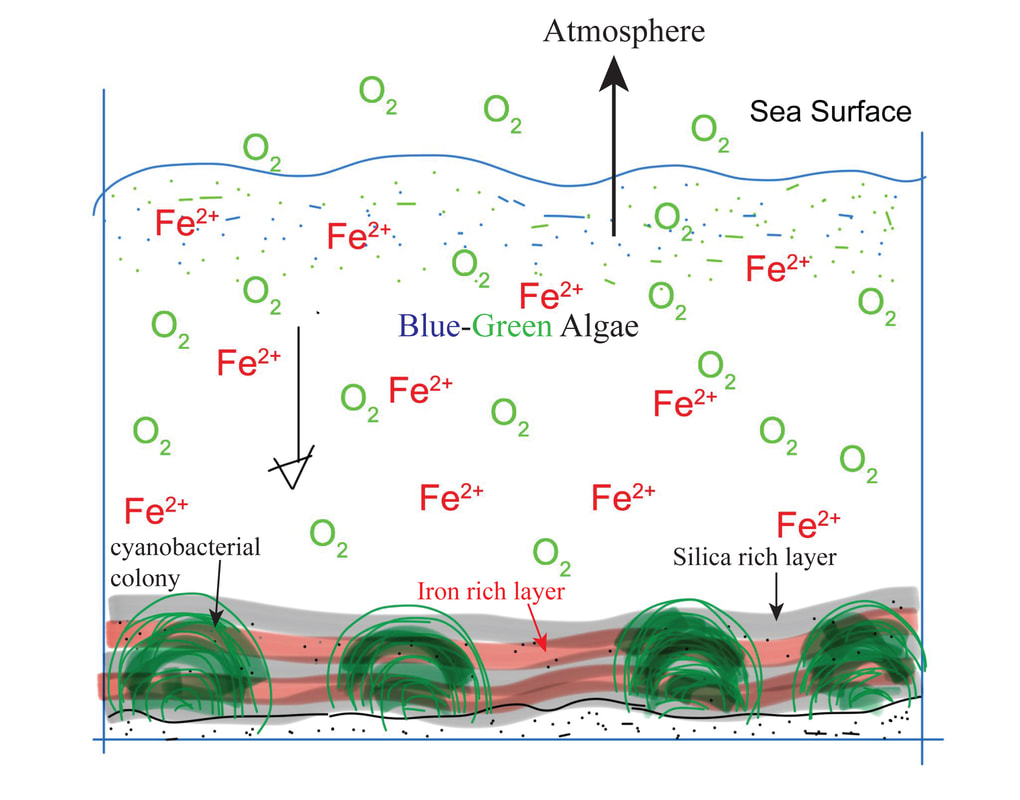
Author: Anupam Atmospheric oxygen, an essential component for all multicellular life forms. The present-day atmosphere has around 21% (by volume) of oxygen, but many of us have limited knowledge on the history of atmospheric oxygen evolution; was that the same as the present-day or different? It will be interesting to know the past concentration of atmospheric oxygen level and how that evolved. We cannot measure the past atmospheric oxygen level directly, but indirectly, using some evidence known as proxies, geoscientists have figured out when and how atmospheric oxygen evolved. There was no free oxygen gas in the Earth's atmosphere at ~4.5 billion years ago. Subsequently, with time atmospheric oxygen reached the present-day level. Two major pulses have been identified during this growth process, the initial one was ~2.5 to 2.2 Ga (billion years), and the second one ~540 Ma (millions of years) (Fig. 1). During this time oxygen level changed (increased) suddenly (sudden with respect to geological time). Entire geological time can be classified broadly into four major segments/stages based on the availability of atmospheric oxygen levels. Stage-1, 4.5 to 2.5 Ga, probably no free oxygen in the atmosphere; Stage-2, 2.5 to 1.8 Ga, O2 produced by cyanobacteria but almost entirely used to reduce seabed rocks and ocean water, part of this time stage is also called as great oxidation event (GOE ~2.4 to ~2 Ga) first step towards the oxygenation of Earth's surface. Stage-3, 1.8 to 0.54 Ga, O2 started coming out from ocean water and absorbed by land surfaces, most probably the formation of the Ozone layer; still, the level of O2 is pretty low than present day, Stage-4, the last one is from 540 Ma to present-day, when the atmospheric oxygen level reached to the present day O2 level (Fig. 1). Free oxygen started accumulating within the ocean water column after the proliferation of early life, i.e., the single-celled organisms, like cyanobacteria, blue-green algae, capable of photosynthesis. Subsequently, free oxygen started reacting with different elements dissolved in seawater and formed element-complexes like iron oxides of higher oxidation state from the earlier lower state. Ancient rock records help us to understand these complex processes. Banded Iron Formation (BIF) is one of those kinds. BIF is a type of sedimentary rock formed due to chemical precipitation from seawater (Fig. 2). These sedimentary rocks are commonly found during Archean to early Paleoproterozoic time; the typical age range is ~2.5 to ~1.8 Ga. BIF is made up of alternate layers of silica /chert-rich and iron-rich materials (Fig. 2). These layering can extend from 10s to 100s of km in length. Relatively young: ~800 to ~600 Ma (millions of years) old BIFs are also present, although they are not very common. Moreover, these relatively young iron formations lack the characteristic banding of alternating silica-rich and iron-rich layers. Depending on the ionic state (redox state) Fe (Fe2+ and Fe3+) behaves differently on an aqueous system. The iron of Fe2+state is soluble in water, where the Fe3+, is insoluble in water. Iron in oxygen-starved oceans of the Archean time was in a ferrous state (Fe2+). Due to the photosynthesis of prokaryotes, oxygen concentration started increasing in the ocean water column, forming iron oxides of Fe3+ oxidation state (like hematite (Fe2O3), magnetite) from Fe2+ state. These Fe3+oxides (insoluble) precipitated to the ocean bottom and formed an iron-rich layer. With time when the entire iron of seawater was consumed, then silica (from the water column) started forming silicon dioxide (insoluble), and that formed a new layer of silica-rich material over the iron-rich layer, and excess O2 started coming into the atmosphere from seawater (Fig. 3). Still, there was not sufficient free oxygen in the atmosphere, so due to continental weathering, iron (iron in Fe2+ state) continuously reached seawater from continental sources, which again increased iron (Fe2+) concentration in the sea. Although the seafloor-hydrothermal systems act as a significant source of dissolved iron into the oceans, the behavior of this dissolved iron still depended on the O2 content of the ocean water. Further, freshly added dissolved iron (Fe2+) formed another new layer of iron-rich materials over the pre-existing iron and silica-rich layer (Fig. 3) in a cyclic manner and formed BIF*, which continued until the atmospheric oxygen reached a sufficient level to oxidize iron-rich materials (Fe2+) within continents during the weathering process and cut the supply of Fe2+ from continental sources to seawater. So, after a particular geological time, BIF formation stopped, and they never came back. Their abundance in Precambrian (geological time ~4.6 to 0.54 Ga) successions alone are used to argue for an atmosphere devoid of free oxygen. Measuring the age of the BIFs, most of the BIF is from a similar age range ~2.5 to 1.8 Ga; we can pinpoint the time from when atmospheric oxygen level started to increase. Similarly, the sulfur isotope ratio is also a good indicator of atmospheric oxygen evolution. Sulfur has four naturally occurring stable isotopes 32S, 33S, 34S, and 36S. They fractionate differently due to their mass difference if they get any external energy supply. Naturally, lighter isotopes will have higher vibrational energy so that they will go out of the system first during any process like evaporation, condensation, or diffusion. This process is called mass-dependent isotopic fractionation as it depends on the isotopic mass. The temporal trend of isotopic mass difference Δ33 of sulfur (mass difference between 34S and 33S) of different geological materials like pyrite (iron sulfide), barite (barium sulfate), of different ages, starting from the Archean to recent, shows two broad trends. The Δ33S pyrites in sediments deposited post-GOE (~2.4 Ga) have values close to zero (Δ33 = 0) that indicates the mass-dependent fractionation (MDF) of sulfur isotopes. But pyrites older than 2.4 Ga are showing a random relation with time (Fig. 3) and Δ33 is not zero that indicates mass-independent fractionation (MIF). Still, the exact mechanism of mass-independent fractionation is unknown, but it may be due to the dissociation of SO2 by the photochemical reaction. It is believed that the initial rise in atmospheric oxygen level formed the Ozone layer, which prevented UV rays and other high-energy particles from coming in from the extraterrestrial radiations, thereby controlling the mass-independent fractionation of sulfur isotopes. 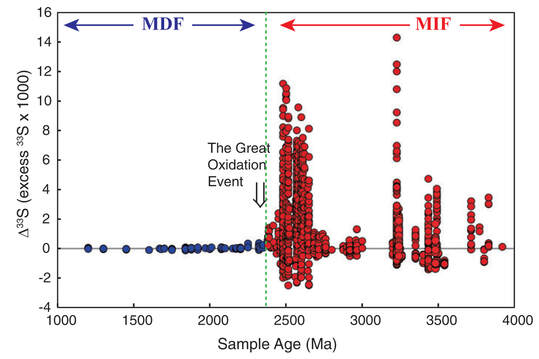 Figure - 4: Temporal distribution of sulfur isotope data evidence for the rise of atmospheric oxygen with the end of SO2 photolytic destruction. Blue and Red circles represent younger and older than ~2.4 Ga, respectively along with that the time of MIF and MDF are also marked (Credit: James Farquhar#) Finally, it can be concluded that the most notable change in atmospheric oxygen evolution happened around the Archean-Proterozoic boundary, ~ 2.5 – 2.2 Ga, which was the first rise of oxygen level in the atmosphere, which reached the present level with another second pulse at around ~540. Probably the cyanobacteria, blue-green algae, were responsible for this evolution. Anupam thanks Mr. Achyuth Venugopal (IISER Pune) for reviewing the initial draft and valuable comments.
*In reality, the formation of BIF is much more complex, and several hypotheses try to explain these enigmatic earth system processes. An in-depth discussion is beyond the scope of this blog post. #Farquhar J. (2018) Sulfur Isotopes. In: White W. (eds) Encyclopedia of Geochemistry. Encyclopedia of Earth Sciences Series. Springer, Cham. https://doi.org/10.1007/978-3-319-39193-9_74-1
0 Comments
Leave a Reply. |



 RSS Feed
RSS Feed
